Article Text
Abstract
Background and aims Patients infected with the SARS-CoV-2 usually report fever and respiratory symptoms. However, multiple gastrointestinal (GI) manifestations such as diarrhoea and abdominal pain have been described. The aim of this study was to evaluate the prevalence of GI symptoms, elevated liver enzymes and mortality of patients with COVID-19.
Methods A systematic review and meta-analysis of published studies that included a cohort of patients infected with SARS-CoV-2 were performed from 1 December 2019 to 15 December 2020. Data were collected by conducting a literature search using PubMed, Embase, Scopus, and Cochrane according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines. We analysed pooled data on the prevalence of individual GI symptoms and elevated liver enzymes and performed subanalyses to investigate the relationship between GI symptoms/elevated liver enzymes, geographical location, mortality, and intensive care unit (ICU) admission.
Results The available data of 78 798 patients positive for SARS-CoV-2 from 158 studies were included in our analysis. The most frequent manifestations were diarrhoea (16.5%, 95% CI 14.2% to 18.4%), nausea (9.7%, 95% CI 9.0% to 13.2%) and elevated liver enzymes (5.6%, 95% CI 4.2% to 9.1%). The overall mortality and GI mortality were 23.5% (95% CI 21.2% to 26.1%) and 3.5% (95% CI 3.1% to 6.2%), respectively. Subgroup analysis showed non-statistically significant associations between GI symptoms/elevated liver enzymes and ICU admissions (OR=1.01, 95% CI 0.55 to 1.83). The GI mortality was 0.9% (95% CI 0.5% to 2.2%) in China and 10.8% (95% CI 7.8% to 11.3%) in the USA.
Conclusion GI symptoms/elevated liver enzymes are common in patients with COVID-19. Our subanalyses showed that the presence of GI symptoms/elevated liver enzymes does not appear to affect mortality or ICU admission rate. Furthermore, the proportion of GI mortality among patients infected with SARS-CoV-2 varied based on geographical location.
- COVID-19
- gastrointestinal tract
- liver
This is an open access article distributed in accordance with the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited, appropriate credit is given, any changes made indicated, and the use is non-commercial. See: http://creativecommons.org/licenses/by-nc/4.0/.
Statistics from Altmetric.com
Introduction
In December 2019, China was faced with a new strain of coronavirus, novel coronavirus (2019 nCov). Within a short period of time, it manifested into a full pandemic.1 It was first noticed by the innumerable cases of pneumonia that suddenly surged among local inhabitants in the province of Wuhan.2 Soon, the virus was detected through sequencing, leading to it officially being renamed severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) by the International Committee on Taxonomy of Viruses.3 The disease caused by SARS-CoV-2 was allocated the title of COVID-19 or ‘coronavirus disease’.3 Coronaviruses in general are single-stranded RNA viruses falling under the family of Coronaviridae, which also include Middle East respiratory syndrome coronavirus (MERS Cov) and SARS (SARS-CoV).4 By the end of December 2020, more than 81 million cases of COVID-19 have officially been confirmed worldwide, and mortality from COVID-19 was more than 1 798 050 deaths worldwide.5 In addition, new variants of SARS-CoV-2 have been discovered in the UK, South Africa, and other regions around the world.6
It has been established that the transmission of SARS-CoV-2 occurs from person to person through the upper airway tract (droplet infection) or through direct contact.7 The virus can also be detected in saliva, urine, gastrointestinal (GI) tract and possibly through airborne spread.8 9 The spectrum of symptoms attributable to SARS-CoV-2 includes fever, cough, myalgia, fatigue, and, to a lesser extent, headache. Patients may also be asymptomatic.10–12 Diarrhoea, nausea and vomiting, as well as liver involvement have all been reported in the literature.13 14 In fact, GI involvement is plausible, given that ACE2, the major human cellular receptor for the SARS-CoV-2, is expressed in the GI tract, as well as in liver cells.15 We thus conducted a systematic review of published GI symptoms and elevated liver enzymes associated with COVID-19 on the basis of disease severity, mortality, intensive care unit (ICU) admission, and geographical region. This will aid in understanding the magnitude of involvement of the GI tract and liver in patients with COVID-19.
Methods
Search strategy
A systematic review was conducted using PubMed, Scopus, Cochrane, and Embase databases. Medical literature searches for human studies were performed from 1 December 2019 up to 15 December 2020. The key terms used for the literature search were ((“COVID-19” OR “COVID 2019” OR “severe acute respiratory syndrome coronavirus 2” OR “severe acute respiratory syndrome coronavirus 2” OR “2019 nCoV” OR “SARS-COV2” OR “2019nCoV” OR (“severe acute respiratory syndrome coronavirus 2” OR “SARS-COV2” AND “gastrointestinal” AND (“manifestations” OR “clinical characteristics”) OR (“gastrointestinal tract” OR (“gastrointestinal’ AND “tract”) OR “gastrointestinal tract” OR (“gi” AND “tract”) OR (“fatality” or “Mortality”). In addition, a manual search of all review articles, editorials and retrieved original studies was also performed. All procedures used in this meta-analysis were consistent with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines and prespecified protocol, which described our method and analysis before data collection was initiated (see online supplemental material PRISMA checklist).
Supplemental material
Selection criteria and data extraction
Data were independently extracted by two investigators (MS and FA) and any discrepancies between the two authors were resolved through discussion. Inclusion and exclusion criteria were defined prior to the literature search. The inclusion criteria were (1) study type: case reports/case series (including chart reviews), prospective/retrospective cohort studies, case–control studies, cross-sectional studies and randomised controlled trials; (2) patient population: adult patients with COVID-19, inpatient or outpatient setting; and (3) outcome measured: at least one reported GI symptom or elevated liver enzyme, number of patients admitted to ICU, and number of deaths reported. In addition, systematic reviews and meta-analyses were also reviewed for any relevant studies.
For the purpose of this study, elevated liver enzyme defined as aspartate aminotransferase (AST) or alanine aminotransferase (ALT) value above the upper limit of normal of each study laboratory reference range. Furthermore, overall mortality was defined as the proportion of deaths among identified confirmed COVID-19 cases in all studies that reported it. The number of deaths among patients experiencing GI symptoms/elevated liver enzymes was extracted and referred to as GI mortality.
Exclusion criteria were (1) review, opinion, abstracts from conferences, editorials, commentary articles, and review articles; (2) studies without data for retrieval; (3) duplicate studies; (4) asymptomatic patients with COVID-19; and (5) studies that did not report GI symptoms.
Data extraction was performed using Microsoft Excel. The following data were extracted:
Study: author, journal, date, country, number of patients, and study type.
Patients characteristics: mean age, ethnicity, gender, and comorbidities.
Number of reported deaths in all studies.
Number of patients admitted to the ICU.
Number of patients who experienced the following GI symptoms/elevated liver enzymes: abdominal pain, diarrhoea, nausea, anorexia, loss of taste, AST or ALT above the upper limit of normal of each study laboratory reference range.
Risk of bias and certainty of evidence
The Methodical Index for Non-randomized Studies (MINORS)16 was used to assess bias risk. In addition, risk of bias was assessed based on four domains: selection, ascertainment, causality, and reporting. An overall judgement of risk of bias was made based on factors deemed to be most critical for the systematic review (selection criteria, ascertainment of outcome, and follow-up duration).
Statistical analysis
Our primary analysis focused on assessing the weighted pooled prevalence of GI symptoms/elevated liver enzymes in patients with COVID-19 infection, occurring any time during the course of illness. We also conducted subanalyses that looked at the association between GI symptoms/elevated liver enzymes and mortality as well as ICU admission. Categorical variables were described as count (%). Continuous variables were described using mean (SD) if they are normally distributed, median (IQR) if they are not. We pooled the single-arm event rates using a random effects method, and we measured heterogeneity within our studies using the I2 statistic. Subanalyses were described and tested using ORs and 95% CIs to determine statistical significance. STATA V.16 was used to calculate ORs and their respective 95% CI and to create Forest and box plots.
Sensitivity analysis
To examine the effect of the quality of studies on our results, we performed a sensitivity analysis on the prevalence of GI symptoms/elevated liver enzymes by excluding low-quality studies. To do so, we used the modified Newcastle-Ottawa Quality Assessment Scale for non-randomised studies.17 A study with a score of 0–3 was classified as a low-quality study. On the other hand, studies that scored 4 or above were included in the analysis.
Results
Research selection and quality assessment
Overall, 158 studies (online supplemental table 1) from 3175 potentially relevant citations were included in the analysis (figure 1). Most of the included studies were single arm only; very few studies included comparator groups. Furthermore, outcome assessors in all 158 studies were not blinded. Both inpatient and outpatient studies were included. The risk of evidence imprecision was rated as very serious, given that the included studies were all observational studies. Overall, all included studies were rated as having very serious risk of bias because they lacked a control group and had a high risk of confounding and selection bias (online supplemental table 2).
Flow diagram for study selection.
Clinical data
This systematic review included 158 studies2–4 8 12–14 16 18–147 with a total of 78 798 patients that tested positive for SARS-CoV-2 and were included in the analysis. The mean patient age was 55.6 years (±14, 95% CI 48 to 57.3) and 45.2% of the patients were men. Most patients had several comorbidities, the most common being hypertension (28.7%, 95% CI 21.3% to 29.1%), diabetes mellitus (17.4%, 95% CI 13.0% to 19.2%), and cardiovascular diseases (15.7%, 95% CI 13.3% to 17.1%). GI symptoms included nausea, vomiting, abdominal pain, loss of taste, anorexia and diarrhoea (figure 2). Heterogeneity statistic I2 is 95%, which signifies a significant heterogeneity among our studies. The most common reported manifestation among GI symptoms/elevated liver enzymes was diarrhoea (online supplemental figure 1). Specifically, GI symptoms/elevated liver enzymes of patients infected with SARS-CoV-2 are diarrhoea (16.5%, 95% CI 14.2% to 18.4%), nausea (9.7%, 95% CI 9.0% to 13.2%), anorexia or loss of appetite (1.6%, 95% CI 1.2% to 5.1%), vomiting (1.5%, 95% CI 5.1% to 8.0%), abdominal pain (4.5%, 95% CI 3.1% to 7.3%), loss of taste (1.3%, 95% CI 1.1% to 4.1%), and elevated liver enzymes (5.6%, 95% CI 4.2% to 9.1%) (online supplemental table 3).
Box plots showing the distribution and proportion of GI symptoms/elevated liver enzymes in patients with COVID-19. GI, gastrointestinal.
Sensitivity analysis
The sensitivity analysis included 52 studies (online supplemental tables 4 and 5). The results did not differ from our main analysis. Among the GI manifestations experienced by patients with COVID-19, diarrhoea (16.6%, 95% CI 12.1% to 17.3%) was still the most common symptom, followed by nausea (9.9%, 95% CI 8.2% to 11.7%). The proportion of patients experiencing loss of taste was 4.7% (95% CI 3.8% to 5.9%). The percentage of patients experiencing elevated liver enzymes was 1.9% (95% CI% 1.3% to 3.4%).
Mortality and geographical variation
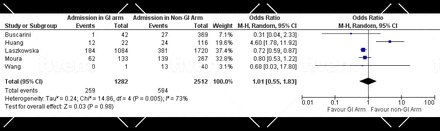
A total of 83 studies reported mortality. Of those, 82 studies reported mortality as the number of deaths at the time of the study. Only one study reported mortality as death over 30 days.143 The overall prevalence of overall mortality and GI mortality were 23.5% (95% CI 21.2% to 26.1%) and 3.5% (95% CI 3.1% to 6.2%), respectively (online supplemental tables 6 and 7). The subgroup analysis included eight studies19 20 57 110 136 139 141 143 that directly compared the number of deaths in patients with and without GI symptoms/elevated liver enzymes. In this analysis, the number of patients who experienced GI symptoms/elevated liver enzymes and those who did not were 1593 and 3321, respectively. The results showed that patients with GI symptoms/elevated liver enzymes were not more likely to die compared with those who did not, with a statistically insignificant pooled odds of patients of 1.01 (95% CI 0.46 to 2.25) (figure 3).
Forest plot of GI mortality in patients with COVID-19, showing no significant difference in the pooled odds of patients with GI symptoms/elevated liver enzymes and those without. GI, gastrointestinal.
Moreover, out of the 158 studies, a total of 42 studies reported mortality in patients with GI symptoms/elevated liver enzymes based on their geographical location (figure 4). This analysis showed that 44 out 4946 patients (0.9%) in China died (95% CI 0.5 to 2.2), whereas 192 out 1783 patients (10.8%) in the USA died (95% CI 7.8 to 11.3). In addition, 2 out of 9 patients (22.2%) in Italy died, while 28 out of 400 patients (7%) in Brazil died. Furthermore, three studies from Taiwan, Korea, and Japan reported zero GI mortality (table 1).
Forest plot of GI mortality in patients with COVID-19 who are experiencing GI symptoms/elevated liver enzymes in three different countries. GI, gastrointestinal.
GI mortality by geographical location
ICU admission rate
Five studies22 85 139 141 148 reported differences in ICU admissions among patients manifesting GI symptoms/elevated liver enzymes and patients who did not. The total number of patients with GI symptoms/elevated liver enzymes who were admitted to the ICU was 1282, and the number of patients who did not experience GI symptoms/elevated liver enzymes and were admitted to the ICU was 2512. No statistically significant difference in ICU admission rate was noted between those who experienced GI symptoms/elevated liver enzymes and those who did not. The pooled proportion was 1.01 (95% CI 0.55 to 1.83) (figure 5).
Forest plot showing odd ratio (OR) of intensive care unit admissions in patients with COVID-19 with and without GI symptoms/elevated liver enzymes. GI, gastrointestinal.
Discussion
This meta-analysis of 78 798 patients with COVID-19 found that GI symptoms/elevated liver enzymes are common in patients infected with SARS-CoV-2. Subgroup analysis found that no association between the presence of GI symptoms/elevated liver enzymes and mortality or ICU admission, which is similar to the finding of other meta-analyses.149 150
GI symptoms including abdominal pain, diarrhoea, nausea, vomiting, loss of appetite, loss of taste and elevated liver enzyme are among the presenting symptoms or laboratory abnormalities of SARS-CoV-2 infection found in this study. Diarrhoea was the most common GI symptom; this is particularly important because previous studies have shown that patients with diarrhoea on presentation have a higher stool RNA positivity and viral load than those without.22 36 151 One study showed that 44 of 153 patients with COVID-19 tested positive for the virus in the stools.148 In addition, a report of a patient with COVID-19 with positive faecal but negative pharyngeal and sputum viral tests has been described.33 Moreover, a meta-analysis concluded that SARS‐CoV‐2 is commonly present in stool samples or anal swabs in which the virus can persist for a long period after respiratory samples become negative and that the virus may be viable.152 This may imply that faecal oral route is a possible route of SARS-CoV-2 transmission.
The possibility of faecal oral transmission of SARS-CoV-2 emphasises the importance of frequent and proper hand hygiene. This is important in every clinical setting, but especially in low-resource areas with poor sanitation.38 Intuitively, proper handling of the excreta of patients with COVID-19 should still be strongly enforced, and sewage from hospitals should also be properly disinfected. The presence of the virus in the digestive tract also raises the concerns of COVID-19 infection in patients with established GI conditions, as well as potential faecal microbiota transplant donors.148 Nevertheless, the unknown effect of COVID-19 on patients with pre-existing GI diseases and its influence on treatment and outcome is a cause for concern. These implications warrant further investigation. The American Gastroenterological Association and joint society recommend the use of enhanced personal protective equipment, including the use of N95 (or N99) masks instead of surgical masks, for healthcare workers performing upper or lower GI procedures regardless of COVID-19 status.35
It is believed that the prevalence of GI symptoms is underestimated because the majority of studies included in our study reported GI symptoms only on the day of admission but not throughout the disease course. Furthermore, many earlier studies did not report on other GI symptoms except for diarrhoea.22 Based on these findings, clinicians must be aware that digestive symptoms, such as diarrhoea, may be a presenting feature of COVID-19 that can arise before respiratory symptoms and, on rare occasions, may be the only presenting manifestation of COVID-19.33
The pooled analysis showed that the overall mortality and GI mortality were 23.5% and 3.5%, respectively. However, it is important to emphasise that reporting of COVID-19 mortality in each country varies.153 Some countries do not depend on the availability of confirmed laboratory tests; instead, both probable and confirmed cases are used when calculating COVID-19 mortality.154
In this meta-analysis, a subanalysis of mortality in patients with GI symptoms/elevated liver enzymes varied between countries. This difference in GI mortality can be attributed to several reasons. Differences in reporting cases, case definition, and the mortality measure used might have a great role in this geographical variation. The available mortality data mostly reported as case fatality rate, which measures the number of deaths out of all confirmed cases.155 Furthermore, using case fatality rate is influenced by reporting and testing strategies in each country, where countries that do not have good reporting or intensive testing might miss a lot of confirmed cases and eventually overestimate mortality.153 In addition, it is well known that comorbidities increase the risk of death from COVID-19,154 and countries with the highest burden of chronic diseases had the highest COVID-19 mortality. Small sample size of the published GI mortality reports of some countries is another factor that can lead to inaccurate presentation of the actual GI mortality.
Our study did not show higher GI mortality among patients manifesting GI symptoms/elevated liver enzymes. However, any possible true difference in mortality may be worth further investigation among better defined patients with COVID-19 subgroups with GI symptoms/elevated liver enzymes because one study showed that prevalence of severe disease was more common in patients who had GI symptoms than those who did not.156 Our meta-analysis did not find a statistically significant association between patients with GI symptoms/elevated liver enzymes and ICU admission. However, to investigate such an association, it is important to consider other causes of elevated liver enzymes in patients admitted to ICU such as sepsis, hypoperfusion, hepatotoxic drugs, and parenteral nutrition.157
Strengths and limitations
Our study has several strengths. This is one of the more recent meta-analyses that summarises the literature on COVID-19 and the prevalence of overall and individual GI manifestations.149 158–161 The large patient population and comprehensive inclusion of 158 studies allow a more precise estimation of the prevalence of GI symptoms/elevated liver enzymes associated with COVID-19. Moreover, our search included studies over 1-year period, from 1 December 2019 up to 15 December 2020, which makes it more up-to-date and more inclusive of the recent evidence. Furthermore, our meta-analysis included studies from different countries and regions.
This study, however, is subject to some limitations. Most of the studies we base our analyses on are observational, single-arm cohorts. The lack of control groups and comparison arms can lead to bias due to confounding. Additionally, regarding mortality among patients with COVID-19, most studies reported mortality at the time of the study. In other words, studies did not report mortality over a specific period of time. Furthermore, most studies reported patients with COVID-19 who have been admitted to hospital, who are more likely to have severe disease, resulting in under-representation of patients with milder disease.
Conclusion
In this meta-analysis, we summarise the recent reports of GI symptoms/elevated liver enzymes among patients infected with SARS-CoV-2. GI symptoms/elevated liver enzymes are commonly observed in patients with COVID-19; therefore, clinicians should be aware that diarrhoea and nausea can be the only manifestations of patients with COVID-19. Our subanalysis showed that GI mortality among patients infected with SARS-CoV-2 varied based on geographical location. We also could not find a statistically significant association between ICU admission in patients with GI symptoms/elevated liver enzymes compared with those without GI symptoms/elevated liver enzymes. However, further investigation is warranted to better assess this possible association.
Acknowledgments
We acknowledge and thank all health care workers and front liners around the globe for their hard work and sacrifices made during this pandemic.
References
Supplementary materials
Supplementary Data
This web only file has been produced by the BMJ Publishing Group from an electronic file supplied by the author(s) and has not been edited for content.
Footnotes
Twitter @drmohamadshehab
Contributors MS: study concept and design, acquisition of data, analysis and interpretation of data, drafting of the manuscript, critical revision of the manuscript for important intellectual content, statistical analysis, and submission of the manuscript. FA: acquisition of data, analysis and interpretation of data, drafting of the manuscript.SS: acquisition of data and drafting of the manuscript. DA: acquisition of data and drafting of the manuscript. AB: critical revision of the manuscript for important intellectual content, statistical analysis, study supervision; he is also responsible for the overall work as a guarantor.
Funding The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests None declared.
Patient consent for publication Not required.
Provenance and peer review Not commissioned; externally peer reviewed.
Data availability statement All data generated or analysed during this study are included in this published article (and its supplementary information files).
Supplemental material This content has been supplied by the author(s). It has not been vetted by BMJ Publishing Group Limited (BMJ) and may not have been peer-reviewed. Any opinions or recommendations discussed are solely those of the author(s) and are not endorsed by BMJ. BMJ disclaims all liability and responsibility arising from any reliance placed on the content. Where the content includes any translated material, BMJ does not warrant the accuracy and reliability of the translations (including but not limited to local regulations, clinical guidelines, terminology, drug names and drug dosages), and is not responsible for any error and/or omissions arising from translation and adaptation or otherwise.